 |
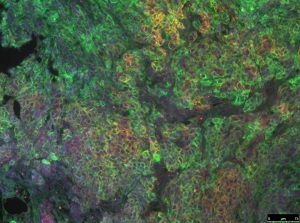
| Cells in a pancreatic tumor, with yellow areas showing where C-MET and CD44v6 are together--Courtesy of KIT |
Working with the knowledge that the CD44v6 protein acts as a co-receptor for receptor tyrosine kinases like MET and VEGFR-2 that drive pancreatic cancer, a group of German investigators at the Karlsruher Institut fur Technologie (KIT) say they've successfully tested a new peptide approach in mice that throws up a hurdle that can blunt and perhaps even stop the development of the disease.
They used tiny segments of the CD44v6 protein--called v6pep--to interfere with the three-dimensional structure of the target protein, which is what they assume led to the therapeutic effect in mice.
The investigators say that not only did they track a shrinkage of tumors and an inhibition of metastasis, they also say that the metastases regressed in their animal models. The work was published in the journal Gastroenterology.
It bears repeating here that mouse models can be notoriously inefficient when it comes to predicting what will work in humans. But the researchers believe that if their peptide segment theory is right, this approach should prove effective in humans.
Having an effect on metastasis in pancreatic cancer is particularly important as most cases aren't diagnosed until the cancer has begun to spread, making this one of the most lethal cancers oncologists have to deal with. And a spinoff of KIT, amcure GmbH, plans to pursue clinical trials in 2016.
"We think that these peptides have a high potential for tumor therapy, above all for the treatment of pancreatic cancer with its aggressive metastases," Véronique Orian-Rousseau of the Institute of Toxicology and Genetics of KIT says.
- here's the release
- read the research abstract