 |
| MannKind CEO Al Mann |
Three years and hundreds of millions of dollars after MannKind was handed its second FDA rejection on Afrezza, the third time proved the charm for the inhaled insulin. The FDA on Friday handed out its blessing for MannKind ($MNKD) to start marketing the drug, which will now set out to see if it can succeed with an inhaled insulin after Pfizer's ($PFE) Exubera failed so spectacularly at the task.
The market is huge. Some 26 million Americans have Type 2 diabetes, threatened by high levels of blood sugar. Valencia, CA-based MannKind--cheered, chaired and financed to a large degree by Al Mann--has long believed that its inhaled insulin offered an easier way for many of those people to control the disease. But after Pfizer had failed to make a go of it, potential major league partners kept their distance, forcing the biotech to dig deep--and borrow more--to finish the last two Phase III studies.
"Afrezza is a new treatment option for patients with diabetes requiring mealtime insulin," said Jean-Marc Guettier, M.D., director of the Division of Metabolism and Endocrinology Products in the FDA's Center for Drug Evaluation and Research. "Today's approval broadens the options available for delivering mealtime insulin in the overall management of patients with diabetes who require it to control blood sugar levels."
Afrezza has been under FDA review off and on for the past 5 years and was rejected for a second time in 2011 after the company switched inhalers while it was under consideration. The writing on the wall for Afrezza finally looked good for the biotech following an overwhelmingly positive FDA panel vote on April 1. The outside experts clearly felt that investigators had provided a positive look at the risk/benefit profile of the treatment. The vote in its favor came despite an internal FDA review that raised fresh questions about risks of bronchial spasms, an impact on lung function as well as the efficacy data on Type 1 patients.
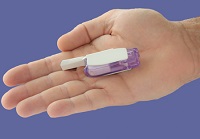 |
| MannKind's Afrezza is delivered through a whistle-shaped inhaler called Dreamboat.--Courtesy of MannKind |
The FDA noted in its release that Type 1 diabetics who use Afrezza will need to continue to use a long-acting insulin. The agency also laid out a risk mitigation plan and requirements for a slate of post-marketing studies to examine risk as well as patients' reactions to the therapy.
Afrezza will carry a label with a boxed warning of acute bronchospasm for patients with chronic lung disease, the risk of hypoglycemia, a decline in pulmonary function, and other potential reactions,
Skeptics have focused plenty of negative attention on MannKind over the years. But back in 2011, MannKind borrowed $370 million to rerun the inhaled drug in Phase III. The same skeptics, though, have downplayed this dry powder's ability to compete against well established injectables, especially after much bigger companies have tried and failed before.
Pfizer pulled Exubera from the market back in 2007, a little more than a year after the agency handed out its endorsement, Jinxed by a high price and hard-to-use inhaler, patients stayed away in droves and predictions of certain blockbuster status had quickly evaporated.
MannKind's response, though, is that Afrezza's formulation and inhaler is different than its predecessors. Now it will have to prove itself all over again. Analysts estimate the treatment should be able to earn about $600 million a year. MannKind's shares slipped about 6% at the close today.
"Approval of Afrezza is an important milestone for MannKind, as today's FDA action validates the years of clinical research and commitment that powered the development of this unique therapy," said Mann in a statement.
- here's the release from the FDA
- here's the release from MannKind