 |
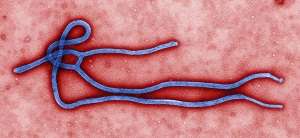
| Ebola virus under an electron microscope--Courtesy of CDC |
When GlaxoSmithKline ($GSK) bought out Okairos last year for $325 million, the platform vaccine technology it acquired was focused on a slate of early-stage development programs covering a range of targets like hepatitis C, malaria and tuberculosis. But in recent weeks the international frenzy over a serious outbreak of Ebola in West Africa has pushed that program to center stage. And today the pharma giant--one of the world's top vaccine players--has joined hands with a global consortium of agencies, nonprofits and investigators to compress a years-long development effort into a matter of weeks as the outbreak worsens.
Initially, GSK officials had sounded distinctly skeptical about whether the vaccine could be ready in time to help contain the outbreak. But this morning there was a distinct can-do attitude in its approach to the crisis.
Using a $4 million grant, Oxford's Adrian Hill will launch a Phase I safety test in about two weeks, once it locks up the necessary paperwork. Hill's study in volunteers will take place alongside separate trials at the National Institute of Allergy and Infectious Diseases in the U.S. And after it's been run through small studies on the healthy volunteers, a green light from regulators would allow investigators to immediately broaden the work to an at-risk population in Gambia and Mali to evaluate whether or not it can contain the virus.
While investigators are testing the vaccine in volunteers, GSK plans to simultaneously ramp up production of 10,000 doses, prepping for a quick vaccination campaign.
If all goes well, GSK says the early-stage study can be wrapped in 3 months--virtual light speed in the pharma industry.
The consortium is gambling on next-gen vaccine technology that was spun out of Merck back in 2007. Inspired by the research of Okairos founder Riccardo Cortese, investigators have been using deactivated chimp viruses to deliver genetic material to cells, triggering a powerful CD8 T cell response--an immune response that can be directed squarely at an invader like Ebola. Most of the attention for Okairos, a 2012 Fierce 15 winner, had been focused on its hepatitis C vaccine work.
The lightning quick effort comes as the WHO notes that the number of people afflicted with the deadly Ebola virus could triple in coming months, noting that 40% of all reported cases have occurred in the past three weeks. The epidemic could ultimately hit more than 20,000 people as health officials try to end the outbreak in the next 6 to 9 months.
GSK R&D chief Moncef Slaoui sounded a note of cautious optimism in his statement.
"Today's announcement shows how private and public partners can pull together to respond to this critical public health emergency," said Slaoui. "Developing a new vaccine is complex with no guarantees of success and it's still early days for our Ebola vaccine candidate. But we are encouraged by progress so far and will do the best we can, along with WHO and our partners, to speed up development and explore ways in which the vaccine could contribute to the control of this or future Ebola outbreaks."
- here's the release