 |
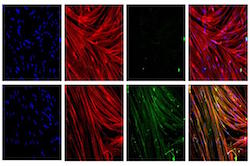
| Top row: skeletal muscle without dystrophin. Bottom row: skeletal muscle with restored dystrophin after CRISPR/Cas9. Nuclei are blue, muscle is red and dystrophin is green. Far right images show overlay of all three--Courtesy of UCLA Broad Stem Cell Research Center |
In December, UT Southwestern researchers used CRISPR/Cas-9 in mouse models to correct the genetic mutation that causes Duchenne muscular dystrophy. Now, UCLA scientists have gone a step further, using the technology to remove the mutation in human cells in a gene therapy that could potentially help 60% of people with the condition.
Duchenne stems from a mutation in the dystrophin gene, which produces a protein of the same name. While there are numerous dystrophin mutations that can lead to Duchenne, the UCLA team zeroed in on the 60% of cases where the mutation occurs in a particular "hot spot" of the gene, the university said in a statement.
They obtained skin cells from patients within this subset and programmed the cells to become induced pluripotent stem cells. They used CRISPR to delete specific parts of the "hot spot," and then turned the muscle cells into cardiac and skeletal muscle cells. When transplanted into mice with the dystrophin mutation, the newly mutation-free cells successfully produced the protein.
This study was the first to correct the mutation in stem cells that could restore functional tissue affected by Duchenne, the statement said.
 |
| (Left to right): Study authors April Pyle (associate professor of microbiology, immunology and molecular genetics and member of the Broad Stem Cell Research Center), Courtney Young (UCLA predoctoral fellow) and Melissa Spencer (professor of neurology in the UCLA David Geffen School of Medicine, co-director of the Center for Duchenne Muscular Dystrophy at UCLA and member of the Broad Stem Cell Research Center) |
"This work demonstrates the feasibility of using a single gene editing platform, plus the regenerative power of stem cells to correct genetic mutations and restore dystrophin production for 60 percent of Duchenne patients," said April Pyle, associate professor of microbiology, immunology and molecular genetics and member of the Broad Stem Cell Research Center.
While the team plans to test this method in animals next, Melissa Spencer, co-director of UCLA's Center for Duchenne Muscular Dystrophy, notes that we likely won't see it tested in people for another 10 years. It is not yet FDA-approved for use in humans.
Despite this caveat, CRISPR represents hope for a potential majority of Duchenne patients. Biotechs such as Sarepta ($SRPT) and BioMarin ($BMRN) have been developing drugs that target the roughly 13% of Duchenne patients who have a particular dystrophin mutation. But just last month, the FDA gave Sarepta's Duchenne drug a scathing review over questionable data a day after the agency formally rejected BioMarin's candidate, citing "contradictory "data.
- read the release