Novo Nordisk ($NVO) has terminated a collaboration deal with Zosano Pharma ($ZSAN), walking away from an agreement that would have paired its late-stage diabetes drug with the latter company's microneedle patch technology.
Novo's decision to nix the deal is tied to "a strategic prioritization" of its diabetes research, according to Zosano, and comes "despite continued progress during the collaboration period." With the cancellation, all of the licensed technologies are returned to Zosano, the company said.
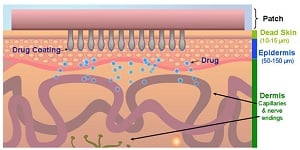 |
| Zosano's microneedle patch delivers drugs via the top layer of skin.--Courtesy of Zosano |
Under the original deal, signed in early 2014, Novo handed over an undisclosed upfront sum to use Zosano's drug delivery technology with its semaglutide, a weekly diabetes treatment now in Phase III. Zosano's proprietary system allows for under-the-skin dosing through a patch dotted with tiny needles, and Novo's candidate, a weekly GLP-1 analog, is designed for subcutaneous delivery. The Danish drugmaker promised up to $60 million in milestone payments for the initial collaboration and as much as $55 million more for each ensuing project.
Novo's exit is a minor blow for Zosano, which is devoting most of its R&D resources to ZP-PTH, a microneedle-delivered hormone treatment designed to treat osteoporosis. Eli Lilly ($LLY), an equity investor in Zosano, signed on to commercialize the Phase II treatment if and when it's approved, promising up to $425 million in milestone payments. Zosano is also working through Phase II development with a treatment that delivers the hormone glucagon to treat severe hypoglycemia and has a preclinical microneedle therapy for migraine.
Zosano, a Johnson & Johnson ($JNJ) spinoff, pulled off a $50 million IPO in January with Lilly's help, and its share value has slipped nearly 30% since.
- read the statement (PDF)