Novartis ($NVS), Juno Therapeutics ($JUNO) and Kite Pharma ($KITE) have made headlines around the world with a new technology for modifying T cells to better attack cancer. But translating clinical promise into a marketable product will likely prove difficult if and when so-called CAR-T therapies win approval.
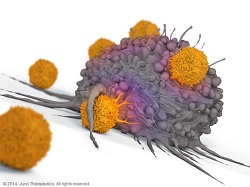 |
| Depiction of a CAR-T cell--Courtesy of Juno |
As Reuters reports, each contender is working to get ahead of the issue by investing in in-house, commercial-scale manufacturing operations, deciding against the more common practice of relying on outsourcers.
Creating therapeutic CAR-Ts requires extracting cells from patients, genetically modifying them and then re-infusing the new material in a process that takes about two weeks per person, according to Reuters. That's a stark contrast to the scalable processes behind the small- and large-molecule therapies that dominate the market for most diseases, and making CAR-T cells for commercialization promises to be a complicated, expensive proposition.
Novartis, expected to be the first to market a CAR-T therapy, has been churning out experimental doses of its product through a facility shared with its partners at the University of Pennsylvania and has acquired a commercial-scale plant from Dendreon, whose own attempts to market a cell therapy ended in bankruptcy. Juno is weaning itself off of contract manufacturing after leasing a Washington plant to crank out its investigational candidate, expecting to bring the facility online next year. And Kite has signed agreements to lease two production hubs, one for making experimental doses and another for commercial manufacturing.
In clinical data released over the past two years, CAR-T therapies have charted stellar results in rare cancers with particularly bleak prognoses, extending patients' lives and delivering complete remissions. Assuming each company can follow through on that promise in late-stage testing, the therapies are likely to win FDA approval, but the cost and complexity of manufacturing doses could limit the immediate market potential of the CAR-T class.
- read the story