 |
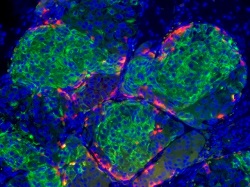
| Insulin-producing beta cells (green) generated from embryonic stem cells--Courtesy of the Melton Lab/Harvard University |
In a notable achievement that could pave the way for less invasive and more permanent treatment options for Type 1 diabetes, scientists from Harvard University have developed a technique that coaxes embryonic stem cells into fully functioning human insulin-producing beta cells.
The method could aid drug discovery efforts and serve as the basis for cell transplantation therapy for the treatment of Type 1 diabetes, which affects an estimated 3 million Americans. The current standard of care for Type 1 diabetics relies on regular injections of insulin to keep their blood glucose levels in check.
Researchers have long sought a way to generate insulin-producing pancreatic beta cells to replace the ones that get destroyed by the immune system in Type 1 diabetes. This destructive immune response causes the body to stop making insulin needed each day.
To restore this function, the Harvard team needed to find the right combination of chemical signals to coax human embryonic stem cells into beta cells, a process called differentiation.
Led by Doug Melton, the researchers studied all the genes that are switched on and off during the normal development of a beta cell in mice, frogs and human material. After identifying these genes, Melton and his colleagues had to figure out a way to manipulate their activity to test which genes are involved in differentiation. They did that by testing hundreds of combinations of inducing agents--small chemicals and growth factors.
Finally, they cracked the code, finding a 6-step procedure that was able to generate hundreds of millions of insulin-producing beta cells that resemble normally functioning beta cells in humans. The authors say this protocol, detailed in the journal Cell, is scalable and could be used to derive the massive quantities of beta cells needed for cell transplantation and pharmaceutical testing.
Investigators are now studying the stem cell-derived beta cells in animal models, including nonhuman primates.
- get the news report from Harvard
- see the study abstract