Boston Scientific has agreed to buy Claret Medical and its neurovascular brain-protection system for $220 million upfront, with potential milestone payments of up to $50 million based on future product reimbursements.
Claret’s Sentinel system uses a net-like device to catch debris knocked loose during transcatheter aortic valve replacements, such as calcium deposits or tissue, as they travel through the aortic arch and the brachiocephalic and carotid arteries, before getting lodged in the brain.
“This acquisition will expand our commercial portfolio to include an important adjunctive offering aimed at improving TAVR patient outcomes,” Boston Scientific’s president of interventional cardiology, Kevin Ballinger, said in a statement. The Marlborough, Massachusetts-based medtech giant also believes the addition could complement the other acquisitions made in its structural and heart rhythm divisions over the past year and a half, as the company continues its shopping spree.
“We also see potential for future use in other left heart and endovascular procedures such as mitral valve repair and replacement, left atrial appendage closure and pulmonary vein isolation ablation procedures for atrial fibrillation,” Ballinger said.
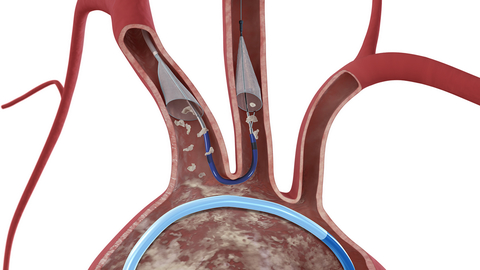
Studies have estimated that approximately 4% of patients experience a stroke within 30 days of a TAVR procedure, the company said. Sentinel received a CE mark in 2014 and FDA clearance in 2017, as the only device cleared to protect patients against the risk of stroke during TAVR procedures. In a clinical trial, Sentinel reduced the incidence of strokes by 63% within the first 72 hours of the procedure, capturing debris flowing toward the brain in 99% of TAVR cases regardless of the type of replacement valve.
RELATED: Boston Scientific to drop $435M on Swiss heart valve maker
In a February conference call with investors, Boston Scientific CEO Michael Mahoney said 2018 could see several acquisitions.
“We do have a pretty active M&A appetite,” Mahoney said, with the company forecasting the availability of $500 million from its cash flow to drive its spending, as it estimated revenues of $9.65 billion to $9.80 billion.
Earlier this month, Boston Scientific acquired Cryterion, makers of a cryoablation device platform for treating atrial fibrillation, for $202 million. The purchase allows the company to offer the delivery of cooling energy in the procedure, which interrupts abnormal electrical signals in heart tissue, in addition to its heat-based radio-frequency technology. Before that, it bought up Securus Medical and its internal body temperature monitors for added safety in ablation procedures.
In January, the company offered $90 million upfront and $450 million in milestones for Millipede’s ring system for mitral regurgitation. The startup’s transcatheter device is being studied in first-in-human trials.
Last November, however, Boston Scientific had to delay the commercialization of its Lotus Edge aortic valve system, following a recall earlier in 2017 after reports of trouble with the locking mechanism. The company said it plans to launch the TAVR device in the U.S. and European markets in 2019.
RELATED: Claret bags $14M to boost access to brain-protection device
Meanwhile, the deal between Santa Rosa, California-based Claret and Boston Scientific is expected to close in the third quarter. In a statement, Claret President and CEO Azin Parhizgar, Ph.D., cited rapid physician adoption of the safety-net device, with it being used on average in 60% of TAVR procedures across 100 centers within its first year.
“Nearly 10,000 patients have been treated worldwide with the Sentinel System, and we are confident that the leadership of Boston Scientific will enable increased momentum and improved patient access to this valuable technology,” Parhizgar said.