A common treatment for atrial fibrillation sees a surgeon inserting a catheter into the heart, where the device emits either heat or cold energy to destroy and scar over the areas of the organ responsible for sending out irregular heartbeat signals. One major risk of those procedures, however, is that the thermal or cryoablation can damage not only other areas of the heart beyond the afib-causing cells but also the nearby esophagus.
That widespread risk has prompted the invention of a new technique, dubbed pulsed field ablation (PFA), that’s meant to be more focused and less damaging. The FDA has yet to clear any PFA devices for use in the U.S., and Boston Scientific, Johnson & Johnson and Medtronic are among the medtech developers vying to snag the first nod—though the latter may have edged ahead with the publication of promising new study results this week.
Medtronic presented the study results at the American College of Cardiology’s annual scientific session in New Orleans on Monday, the same day they were published in the American Heart Association’s Circulation journal.
PFA systems briefly create an electric field that interrupts the irregular electrical signals in the heart that cause afib. Medtronic’s version of the technology, the PulseSelect device, sends out the electrical field from a circular array of nine electrodes surrounding the end of the ablation catheter, which is positioned near the opening of the pulmonary vein.
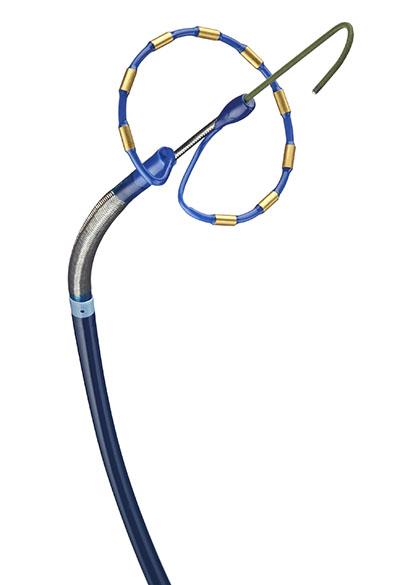
The Pulsed AF pivotal trial of the system spanned nine countries around the world, studying the treatment of 300 participants with either paroxysmal or persistent afib who hadn’t responded to pharmaceutical therapies for the condition.
The researchers judged the efficacy of the PulseSelect system based on whether patients were able to avoid acute procedural failure, recurring arrhythmia and a need for repeat or alternative treatments, among other potential complications and follow-up procedures, within 12 months of the initial PFA procedure. About two-thirds of the paroxysmal afib patients achieved that goal, as did 55% of those with persistent afib—well above the preset thresholds of 50% and 40%, respectively.
Additionally, by the end of the one-year study period, 70% of those with paroxysmal afib and 62% of the persistent afib group were free from recurrence of any atrial arrhythmias, a number that rose to about 80% for both cohorts when looking at freedom from just symptomatic arrhythmias.
In total, the procedures resulted in an adverse event rate of just 0.7% for the entire patient group—a number that Medtronic classified as “one of the lowest adverse event rates of any prior U.S. FDA investigational device exemption trial for atrial fibrillation ablation or any multi-center PFA study.” Plus, the injuries included no instances of esophageal events, pulmonary vein stenosis or phrenic nerve injury, all common complications of standard forms of ablation.
During a call with investors about Medtronic’s third-quarter earnings last month, CEO Geoff Martha said that the then-upcoming trial results marked the first to be reported from a completed investigational device exemption trial for pulsed field ablation technology. He added, “We’re on track to be one of the first companies with a PFA catheter in the U.S. market.”
Among its main competitors in the space is Boston Scientific, which acquired PFA catheter maker Farapulse for $295 million in 2021. A randomized trial of Farapulse’s PFA catheter is also underway, with the final results set to be unveiled later this year.
Hot on their heels is Johnson & Johnson’s Biosense Webster, which launched a clinical trial of its own PFA system in patients with paroxysmal afib early last year. Interim results published last month reported zero primary adverse events among 226 participants, about 71% of whom experienced freedom from recurring symptomatic arrhythmias after 12 months.