of Medtronic
Medtronic has earned a CE mark for the Guardian Connect mobile continuous glucose monitoring (CGM) system for individuals with diabetes who use insulin injection therapy. This is the first CE mark given to any smartphone-enabled CGM system.
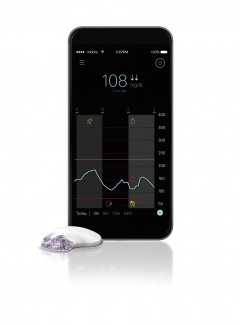
Checking glucose levels on Guardian Connect is as simple as checking text messages or emails on a smartphone, Medtronic ($MDT) explained. Alerts can also be used to notify a patient of high or low glucose levels, determined by a preset measurement threshold.
"Having continuous, real-time access to glucose values and being alerted to important trends and events is key for people with diabetes," said Annette Brüls, president of diabetes service and solutions at Medtronic.
Patients can take advantage of automatic uploads to CareLink therapy management software, so there is no need for bulk data upload by patients or healthcare providers. Brüls noted that the CareLink system “arms healthcare providers, their patients and care partners with actionable data to help improve day-to-day diabetes management and make long-term therapy adjustments."
The system uses a small wearable CGM device that takes glucose measurements every 5 minutes and sends readings to a smartphone app. Users can share data through the aforementioned CareLink system and can also set up SMS text messages to send to care providers.
The system is set to launch country-by-country in the second quarter of 2017 beginning in select countries in Europe, Asia Pacific and Latin America. The smartphone app will initially be available on iOS with an Android version currently in development.
- here's the release
Related Articles:
Abbott snags CE mark for continuous glucose monitor with no finger sticks for kids
U.S. pivotal trial starts for Senseonics glucose monitor implant with data due in June
Medtronic, Qualcomm partner for disposable, connected Type 2 diabetes continuous glucose monitors