Tandem Diabetes Care and TypeZero Technologies have entered into an agreement to bring together TypeZero’s artificial pancreas (AP) tech and Tandem’s t:slim Insulin Pump.
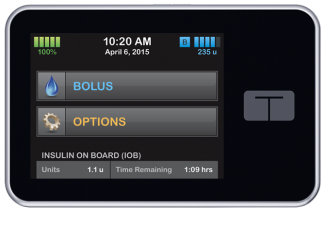
TypeZero’s AP tech has been used in over 28 clinical studies with more than 475 participants. The tech has treat-to-target algorithms that were developed by TypeZero based on initial research done by the University of Virginia.
“We believe that bringing our artificial pancreas technology together with Tandem’s innovative insulin pump platform will revolutionize the way patients manage their diabetes,” Chad Rogers, founder and CEO of TypeZero, said in a statement.
While financial details of the License Agreement have not been disclosed, the announcement did explain that it includes a worldwide, nonexclusive license for the use of TypeZero’s AP tech in future Tandem products. Tandem will also have access to TypeZero’s future AP tech over the next 5 years.
The duo plans to work closely in an effort to expedite clinical research regarding Tandem’s closed-loop AP system. The agreement also noted that a research version of the t:slim pump will be used with TypeZero’s AP tech in the International Diabetes Closed Loop Trial, which takes place later this year. The trial is funded by the National Institutes of Health (NIH).
This trial with the NIH comes off the heels of a workshop regarding TypeZero that took place in July. The NIH planned to discuss the tech and how to make devices safe and efficient for patients.
TypeZero also announced a partnership with Cellnovo Group earlier this year. The two teamed up on a clinical trial for the AP tech, which at the time had already received a $12.8 million grant from the NIH. In the trial, Cellnovo's connected insulin patch pump would be used with TypeZero's inControl AP software and a Dexcom continuous glucose monitor.
- here's the press release
Related Articles:
Bigfoot Biomedical starts clinical trial for artificial pancreas
NIH to weigh in on artificial pancreas development at upcoming meeting
Cellnovo partners with TypeZero in major NIH-backed artificial pancreas trial in Type 1 diabetics