Medtronic has announced FDA approval of the Enlite sensor with iPro2 Professional Continuous Glucose Monitoring (CGM) system.
The sensor is a disposable glucose sensor. It provides the user with more comfort and can be worn for up to 6 days. It is 69% smaller than the previous Medtronic sensor.
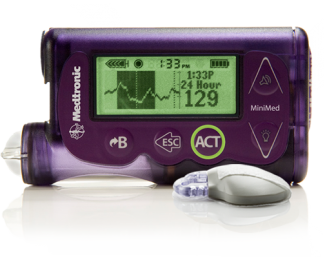
The iPro2 system can provide physicians with insights on how diet, medication and daily activity are affecting the glucose levels of people with diabetes. This offers more insight in less time and helps doctors make more informed decisions on a patient’s diabetes management.
This sharing of information also helps patients to better understand their glucose levels and how those are affected by their lifestyle.
The iPro2 system also offers decision support to care providers through a “Pattern Snapshot” that helps quickly identify and address problem areas.
"We fundamentally believe that to improve outcomes for people with diabetes, physicians need advanced insights to optimize therapy and people with diabetes need to understand the context behind glucose fluctuations to drive behavior change," said Laura Stoltenberg, vice president and general manager of non-intensive diabetes therapies at Medtronic, in the announcement. "Our focus is to continuously enhance our professional CGM solutions and improve patient and physician experience by making the systems easier-to-use through advancements such as the Enlite sensor and our Pattern Snapshot report."
"Both physicians and people with diabetes need easy-to-use systems which can provide actionable information. The new Enlite sensor is easy to insert and comfortable to wear. Having six days of patient data permits robust pattern analysis for more accurate decision support," said Dr. Robert Vigersky, medical director of non-intensive diabetes therapies at Medtronic, in the announcement. "With the insights and reports provided through professional CGM, patients can be shown the factors that are driving glucose fluctuations, so together with their doctor they can develop a plan to improve glucose control."