 |
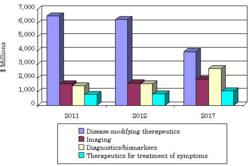
| Global market for Alzheimer's disease, 2011-2017 ($ in millions)--Courtesy of BCC Research |
Anyone trying to understand why biopharma companies regularly burn through hundreds of millions of dollars of their R&D cash on shaky Alzheimer's therapies should check out a new report from BCC looking at the state of the market for dementia drugs. According to analysts, the Alzheimer's drug market peaked last year at $10.2 billion and will now slide 1.5% per year until 2017, when the cash returns will slide to about $9.5 billion.
The reason for the coming slide, they say, revolves around the fact that drug developers have been able to come up with nothing effective to treat Alzheimer's since Aricept was approved 15 years ago. But now that generic alternatives are on the way for Aricept, Exelon and Namenda, the cash flow will start to dwindle significantly.
Of course, any company that manages to get an effective drug through to an approval would be positioned to clean up. Every second a U.S. citizen develops the memory-wasting disease, which now afflicts 5.4 million Americans. And with the world's population aging rapidly, the neurological disease is expected to boom in the elderly, with the patient population swelling from 21 million in 2010 to 53 million in 2050.
Look for the diagnostics and biomarker fields to benefit first as researchers find better ways to diagnose the ailment. And from there drug developers stand to benefit by turning new knowledge about the role of proteins and enzymes into new therapies.
"The market is just waiting for the next big innovation in Alzheimer's disease treatment and prevention," notes BCC. But based on a string of late-stage failures over the past year--for solanezumab, bapineuzumab and Gammagard--there's no telling just how long that wait will last.
- here's the press release