 |
| Courtesy of BioDelivery Sciences |
After years of delay, partners Endo Pharmaceuticals ($ENDP) and BioDelivery Sciences International ($BDSI) won FDA approval for a dissolvable pain treatment, but an on-label warning of serious side effects could dull the drug's effect on the market.
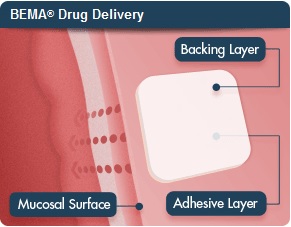
The drug, dubbed Belbuca, is a formulation of the analgesic buprenorphine delivered through a film attached to the cheek, treating chronic pain for 12 hours at a time.
BDSI invented the treatment and pushed into late-stage development on its own before running into a serious setback when the film failed to beat placebo in a 2011 Phase III trial. But Endo had faith in the project, stepping in the following year with a $30 million up-front payment and the promise of as much as $150 million more in exchange for global rights to the drug. And a second Phase III trial, reported in 2014, came through with positive results, laying the path to FDA approval for the partners.
But Belbuca's final label may limit the pair's returns. The drug is indicated for severe chronic pain only among patients for whom non-opioid treatments and immediate-release painkillers are intolerable or ineffective, and it carries a black-box warning describing its potential for abuse and addiction plus risks of fatal respiratory failure.
For BDSI, the approval triggers a $50 million payment from Endo, and the company is entitled to tiered royalties on Belbuca sales once its partner launches the drug in the first quarter of next year. BDSI's shares rose about 18% in premarket trading on Monday.
Belbuca is headed for a crowded analgesic market, but its novel delivery could help it stand out among the oral therapies now dominating the space, according to Nomura analyst Shibani Malhotra, who estimates the drug could bring in peak sales of $500 million.
- read the statement