Eli Lilly ($LLY) offered long-term evidence to show that its amyloid-busting drug solanezumab has a distinct impact on Alzheimer's disease among early-stage patients, pointing to a delayed-start 3.5-year extension study, with a group of patients from the original placebo arm continuing to lag right behind patients who started on the drug at the beginning.
Investigators zeroed in on the performance of patients in the initial placebo arm who were later given solanezumab. Looking over the results on two key measures--ADAS-Cog14 (cognition) and the ADCS-iADL (a daily function assessment)--the placebo group was unable to "catch up" to the patients who were provided solanezumab from the get-go. And that steady gap, Lilly asserts, with the two groups declining on parallel tracks, may well illustrate the disease-modifying effect from the amyloid drug--a landmark event for a field that has seen repeated failures over the years.
Not only did the performance gap persist over a 2-year period, says Eric Siemers, a senior medical director at Lilly, but the placebo patients remained an even step behind, a further indication that they were seeing an effect from the drug.
"Rather than diverging, they remained parallel," says Siemers. Something like that could not be expected to happen by chance.
The news didn't help Lilly's shares, though. The company's stock was down more than 3% in early trading as persistent doubts about the drug's prospects tempered the enthusiasm for Lilly. The pharma's stock declined 2.6% on Tuesday.
There are some big caveats to report, as the study failed to track significant results for two other key measures: A Mini Mental State Examination (MMSE) and the Clinical Dementia Rating scale Sum of Boxes (CDR-SB).
"Results for the ADAS-Cog14 and the ADCS-iADL at the prespecified primary analysis time point of 108 weeks (that is, 28 weeks in the delayed-start period) demonstrated that starting solanezumab early had benefits that persisted after placebo-treated patients began solanezumab 18 months later," Lilly reported in a related study that was posted this morning. "Although the MMSE showed a statistically significant difference between solanezumab and placebo at the end of the placebo-controlled studies, the separation was not statistically significant and the noninferiority criterion was not met at the 108-week time point."
"The lack of statistically significant results on the MMSE and CDR-SB scales warrants some caution in drawing firm conclusions from these analyses," Lilly added. "The safety and efficacy of solanezumab treatment will continue to be evaluated in the ongoing EXPEDITION 3 study in patients with mild AD (NCT01900665), providing an opportunity to replicate these results."
"When it comes to AD, it is hard to know for sure what the different tranches of investors expect, but in our view these results are about as favorable as they could have been," noted Bernstein's Tim Anderson, an analyst who's adopted a consistently upbeat tone when it comes to Lilly's pipeline. "That said, LLY may well suffer from the phenomenon of "buy on the rumor and sell on the news" because from here, there may not be meaningful new news on solanezumab until late 2016 or early 2017 when EXP 3 reports out."
Lilly investigators turned out at the annual Alzheimer's Association International Conference in Washington, DC, to unveil its closely watched study results, which followed a group of patients long after solanezumab failed both of its first Phase III tests.
Lilly--which has gone years without developing a blockbuster medication, with the jury still out on some recently approved drugs--never conceded defeat for solanezumab. When it came time to report Phase III results from two studies in 2013, the company hit hard on a subgroup analysis from the first study--Expedition--in which investigators tracked a significant response among mild, or early-stage patients on cognition, ranked by a rating system called ADAS-cog11. Because of that signal, Lilly regrouped on Expedition 2, changing the endpoint to focus on mild patients' cognitive response to a better, ADAS-cog14 scale.
But that trial failed as well after 80 weeks of treatment.
So Lilly regrouped again, starting a new Phase III study of the drug--Expedition 3--and then enlisting patients from both of the initial Expedition trials into an extension study.
The placebo arm patients paralleled the relentless decline. Here are Lilly's charts on ADAS-cog14 and ADCS-iADL:
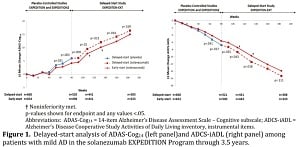 |
| Charts showing results from a delayed-start 3.5-year extension study (Click to enlarge) |
The data are relatively meaningless in terms of Lilly's chances of eventually gaining an approval for the drug, which could easily earn billions of dollars in a market with more than 5 million desperate patients in the U.S. alone. Those hopes will rise or fall with its ongoing Phase III trial. But the new data from the old studies will help boost the crowd inside the Alzheimer's field that believe strongly that the toxic clumps of amyloid, which solanezumab is designed to prevent, is the culprit responsible for extensive brain damage and memory loss.
Lilly hopes to be the first to get a meaningful drug on the market, and the FDA has signaled its willingness to be more flexible in order to make that happen. As of now, Siemers says, Lilly expects to wrap the Expedition 3 study in October 2016, with data to follow soon after, putting Lilly on track to be the first to approach the FDA for an approval.
Alzheimer's has been an extraordinarily frustrating field, with a 99% failure rate over the past decade. But the potential rewards are so big, with billions on the line, that solanezumab remains a key part of Lilly's late-stage case that it can make a comeback after a punishing round of patent losses. And its stock price has surged more than 20% this year, with much of that gain based on anticipation of the extended solanezumab results.
Lilly has also had a significant amount of support for its position among the experts in the field. Solanezumab, which is credited with a fairly good safety profile, was selected for the Anti-amyloid Treatment in Asymptomatic Alzheimer's Disease (A4) prevention clinical trial back in 2013.
"Our confidence level in solanezumab grew after the results were confirmed through an independent, academic analysis by the (Alzheimer's Disease Cooperative Study (ADCS), a clinical-trials network) released in early October," Reisa Sperling of Brigham and Women's Hospital, who is principal investigator for the A4 trial, told Alzforum last year. "We hope that starting treatment much earlier in the disease, before symptoms are present, as well as treating for a longer period of time, will slow cognitive decline and ultimately prevent Alzheimer's disease dementia."
Ironically, the ADCS has been headed by Paul Aisen, a Lilly consultant whose tearful response to the Expedition data helped persuade Lilly to move ahead, according to a report by Bloomberg. Aisen, also a co-lead on the extended study, now is embroiled in a bitter legal dispute with UC San Diego after striking a deal to defect to USC, bringing his research work with him.