With the help of Syncona, the Wellcome Trust's venture arm, Oxford University spinout NightstaRx has $20 million and a plan to develop gene therapies for degenerative eye conditions.
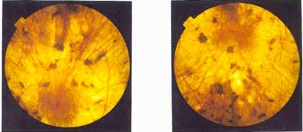 |
| Retina damaged by choroideremia--Courtesy of the Choroideremia Research Foundation |
Based on the work of Oxford's famed Robert MacLaren, NighstaRx's lead candidate is designed to repair light-detecting cells in patients with choroideremia, an inherited form of progressive blindness that stems from a defect in the CHM gene. In a small Phase I study published in The Lancet this month, the treatment, dubbed AAV.REP1, improved the vision of 6 patients with the incurable disease, using a small virus to carry the correct version of the CHM gene into the retina.
Now, with Oxford's go-ahead and Wellcome's financial support, NighstaRx is working through a 12-patient Phase I study of AAV.REP1 to better flesh out its benefits, and the company's founders have recruited former Syntaxin CEO Melanie Lee to lead the way. That $20 million is one of the largest investments in an academic spinout, according to Tom Hockaday, who runs Oxford's research commercialization arm, and the combination of MacLaren's work and Lee's experience with at the helm of an emerging biotech spell a bright future for NighstaRx, he said.
And the company is in it for the long haul, Lee said, using its initial funding to map out a plan for clinical development, manufacturing and eventual regulatory applications for AAV.REP1.
"The initial clinical results for choroideremia gene therapy are very promising, and they give us an indication of what this technology can achieve in the future," Lee said in a statement. "The Wellcome Trust and the University of Oxford are two of the worlds' leading biomedical research organizations, and they have worked closely together to support the program."
- read the announcement