 |
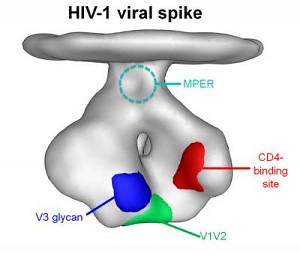
| In this representational image of one of the numerous spikes that jut out of the surface of HIV, the V1V2 region (green) marks where CAP256-VRC26 and other broadly neutralizing HIV antibodies bind.--Courtesy of NIAID |
A new discovery that explains how the body's immune system makes a potent antibody that blocks HIV infection may be an important step toward developing a vaccine against the virus.
Scientists from the National Institute of Allergy and Infectious Diseases analyzed data from the results of a clinical trial of the only experimental HIV vaccine to date that's shown modest success in people. Trial results showed that antibodies that bind to the V1V2 region of HIV's Envelope protein correlated with lower infection rates among those who were vaccinated.
Many researchers believe that an effective vaccine against HIV would work by eliciting powerful antibodies to a specific conserved site on the virus called V1V2, one of a handful of sites that remains constant on the fast-mutating virus.
Along with investigators from Columbia University, the Centre for the AIDS Programme of Research in South Africa and the National Institute for Communicable Diseases in Johannesburg, the NIAID researchers identified an HIV-infected subject who naturally developed V1V2-directed HIV neutralizing antibodies, named CAP256-VRC26, after several months of infection.
Analyses of blood samples donated by the volunteer between 15 weeks and four years after becoming infected allowed scientists to determine the genetic make-up of the original form of the antibody and define the various structures the antibody takes as it mutates toward its full potency.
The study found that after relatively few mutations, even the early intermediate versions of the CAP256-VRC26 antibodies can effectively neutralize many known HIV strains.
The investigators believe that the findings validate the basis for a V1V2-directed HIV vaccine and have begun work on a set of vaccine components designed to produce V1V2 neutralizing antibodies and guide their maturation.
- read the press release
- see the study abstract