 |
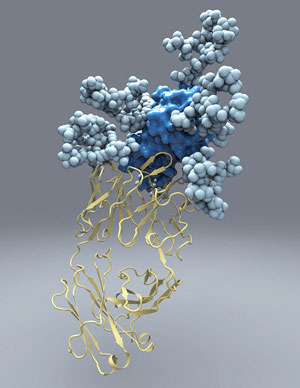
| The new picture of hepatitis C's E2 protein, which the virus uses to infect liver cells, will aid in the design of a vaccine against the disease.--Courtesy of Christina Corbaci, The Scripps Research Institute |
In a discovery that could put a vaccine for hepatitis C closer in sight, researchers have been able to characterize a key protein in the virus that aids infection in the liver.
The structure of the protein, known as E2 envelope glycoprotein, has up until now evaded scientists. But the new, detailed picture of the protein obtained by investigators at The Scripps Research Institute in La Jolla, CA, is an important development toward new, more effective therapies to combat hepatitis C. The findings appeared in the Nov. 29 issue of the journal Science.
"E2 is used to deliver infected cells to patients. It's really the first point of contact between the virus and human cells," Dr. Leo Kong, a Scripps research associate and first author of the study, told FierceBiotechResearch in an interview.
Previously, scientists have found that isolated rare antibodies from patients are able to bind E2 in ways that provide a neutralizing effect on a broad range of viral strains. So the Scripps investigators believe that E2 will provide a likely target--or at least, a starting point--for a successful hep C vaccine.
In the U.S., it's estimated that about 3 million people harbor the hep C virus, which kills more people per year than HIV. But the virus is difficult to detect because it's often harbored for many years before symptoms appear. It's for this reason, as well as currently used antivirals that cause drug resistance, that hepatitis C treatments have had varied success. About 50% to 80% of people treated for hep C are cured, but because the infection can be present with no symptoms, the virus can become a chronic condition, causing liver scarring, cirrhosis or liver cancer.
Available therapies for HCV are not ideal. They come with a host of side effects. Many treatment regimens rely on the use of a combination of several drugs. So far, there is no single regimen used across the board to treat the disease.
Scripps investigators were surprised when after 6 years they finally cracked the code of hep C's E2 protein. They initially believed that it was similar to viral proteins in West Nile and dengue viruses. Instead, they found the structure of E2 in hep C looks--and probably works--differently than what was previously believed.
"What we have now is enough to design immunogens," Kong said. These are antibody-stimulating substances that provoke an immune response in the body--the basis of a future hep C vaccine. Kong and his colleagues are also looking at designing new small molecules that target the E2 protein.
- read the press release
- here's the study abstract