 |
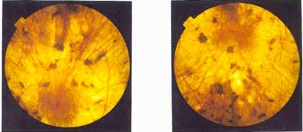
| Retina damaged by choroideremia--Courtesy of the Choroideremia Research Foundation |
An experimental gene therapy has improved the vision of 6 patients in a small Phase I trial in the U.K., exceeding expectations of investigators, according to a new study in The Lancet.
Researchers at the University of Oxford have used a method that replaces a gene in the retina to repair essential light-detecting cells in patients with choroideremia, an inherited cause of progressive blindness. The results are published 6 months after an initial treatment for the disease, which currently has no cure.
Developed by Robert MacLaren of the Nuffield Laboratory of Ophthalmology at the University of Oxford, the gene therapy uses a small virus to carry the CHM gene, which is defective in patients with choroideremia. Once the CHM gene reaches the light-sensing cells in the retina, it spurs the production of protein to stop these retina cells from dying off. Without this crucial protein produced by the CHM gene, pigment cells in the retina of the eye slowly stop working and then die off, shrinking the retina and reducing vision. Choroideremia affects mostly boys, as the mutation occurs in the CHM gene on the X chromosome.
Before the start of the trial, two of the trial participants had excellent visual acuity, two had good acuity and two had reduced acuity. Visual acuity, or the clearness of vision, is measured by reading lines of letters on a sight chart. Six months after the gene therapy operation to one eye, the subjects with excellent and good acuity had the same level of acuity they began with but could see more in the dark when tested. The two patients with reduced acuity had improved vision and were able to read two and four more lines on the sight chart. One patient with reduced acuity at the start of the trial has shown ongoing improvement for two years, and another has shown continued improvement at one year. So far, 9 patients have had one eye treated with the gene therapy in operations at the Oxford Eye Hospital.
"The results showing improvement in vision in the first six patients confirm that the virus can deliver its DNA payload without causing significant damage to the retina. This has huge implications for anyone with a genetic retinal disease such as age-related macular degeneration or retinitis pigmentosa, because it has for the first time shown that gene therapy can be applied safely before the onset of vision loss," said MacLaren in a statement.
For the next part of the trial, MacLaren said his team is increasing the dose of the gene therapy to find what level is needed to halt degeneration.
The trial is funded by the Health Innovation Challenge Fund, a partnership between the Wellcome Trust and the U.K.'s Department of Health.
Although early clinical failures led many to believe that the promise of gene therapy was all hype, there has been renewed interest in the field in recent years, especially with the approval of uniQure's Glybera gene therapy to treat lipoprotein lipase deficiency.
- here's the University of Oxford press release
- see the Lancet abstract