 |
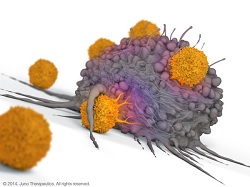
| Illustration of CAR-T technology--Courtesy of Juno |
Seattle's Juno Therapeutics, at work on treatments that train the immune system to better fight cancer, has signed a deal to expand its pipeline of therapies, licensing a complementary candidate developed by the National Cancer Institute.
Under a deal with NCI partner Opus Bio, Juno has paid an undisclosed sum to license an immunotherapy that targets CD22, a protein expressed on B cell leukemias and lymphomas. Juno's work in the field has thus far focused on CD19, another protein common among blood cancers.
Much like the rest of Juno's pipeline, the NCI treatment is a CAR-T immunotherapy, in which scientists remove T cells from a patient's blood and equip them with targeting mechanisms called chimeric antigen receptors (CARs), which seek out and bind to proteins expressed by cancer cells. The resulting cells are reinjected into the patient, at which point they track down malignancies and attack them as they would any commonplace infection.
NCI, in collaboration with Opus, has already begun enrollment in Phase I study testing the candidate in children and adolescents with relapsed/refractory acute lymphoblastic leukemia (ALL) or non-Hodgkin's lymphoma (NHL).
The deal adds a fourth clinical CAR-T candidate to Juno's portfolio of potential therapies. Leading the way is JCAR015, an ALL treatment whose stellar Phase I results led the FDA to grant a breakthrough therapy designation last month, promising an expedited regulatory review. Behind that is JCAR017, in development for ALL and NHL, followed by JCAR014, which is undergoing early studies on various B cell malignancies.
Meanwhile, Juno is queued up to make a splash on Wall Street after filing for a $150 million IPO last month. The biotech has turned heads since its launch last year, raising more than $300 million in venture cash and reporting excellent clinical results for its cadre of cancer treatments, including complete remission rates above 90% in ALL.
Now it's locked in competition with Novartis ($NVS), which is developing a similar--and similarly promising--CAR-T therapy alongside the University of Pennsylvania. And the technology's promise has brought a host of contenders to the table, including oncology heavyweight Celgene ($CELG), which reached out to gene therapy pioneer bluebird bio ($BLUE) to get to work in the space, and Santa Monica, CA's Kite Pharma ($KITE), which grossed more than $140 million in a June IPO on the strength of its approach to T cell modification. Late-comer Johnson & Johnson ($JNJ) has shouldered its way into CAR-T with a $292 million deal signed last month.
- read the release
Special Report: FierceBiotech's 2014 Fierce 15 - Juno Therapeutics