- Data showed atezolizumab can be combined with Zelboraf to enhance the immune system to recognise and destroy cancer cells
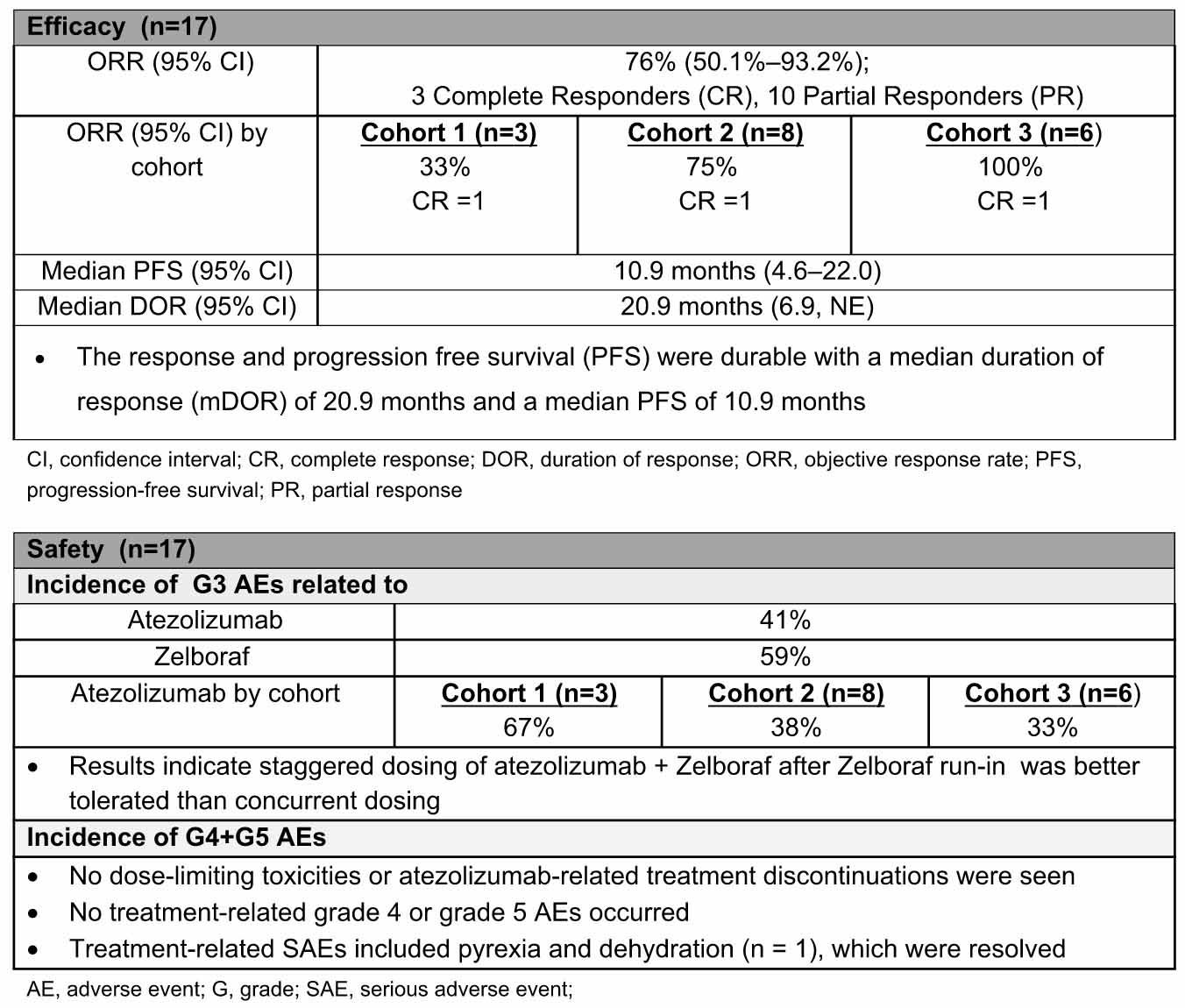
Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced results from a phase Ib study of the investigational cancer immunotherapy atezolizumab (MPDL3280A), used in combination with the BRAF inhibitor Zelboraf® (vemurafenib) for previously untreated BRAFV600 mutation-positive unresectable or metastatic melanoma. Adverse events (AEs) were manageable and generally reversible. The study also showed that the combination resulted in an objective response rate [ORR] of 76% (95% confidence interval [CI:] 50.1%–93.2%) of people (n=17), including three complete responders (CR). The data were presented at the Society of Melanoma Research (SMR) 2015 International Congress.1
"These early efficacy results encourage us to further evaluate combination strategies of atezolizumab and targeted therapies like Zelboraf in people living with advanced melanoma, a disease which is still associated with a poor prognosis", Sandra Horning, MD, Roche's Chief Medical Officer and Head of Global Product Development.
Roche is also investigating a triplet regimen with atezolizumab plus the established targeted therapy combination of Zelboraf and Cotellic® (cobimetinib), a selective MEK inhibitor, in a phase Ib study.
About the phase Ib study of atezolizumab in combination with Zelboraf
- This phase Ib, open-label study aimed to evaluate the safety and pharmacology of atezolizumab in combination with Zelboraf in patients with previously untreated BRAFV600 mutation-positive metastatic melanoma
- 17 patients were evaluable for safety and efficacy at the time of this data cut
- Patients received atezolizumab combined with Zelboraf concurrently (Cohort 1 [C1], n=3) or after a run-in period with Zelboraf alone of 56 days (Cohort 2 [C2]; n=8) or 28 days (Cohort 3 [C3]; n=6)
- Patients were given atezolizumab intravenously every 3 weeks at 20 mg/kg (C1) or 15 mg/kg (or 1,200 mg fixed; C2/C3). Oral Zelboraf was given twice daily at 960 mg during the run-in period and at 720 mg during atezolizumab and Zelboraf combined treatment
- PD-L1 expression was centrally assayed using immunohistochemistry (IHC; SP142 assay)
About BRAFV600 mutation-positive metastatic melanoma
Melanoma is less common, but more aggressive and deadlier, than other forms of skin cancer.2,3 A mutation of the BRAF protein occurs in approximately half of melanomas, and a test can be used to determine who can be treated with a BRAF-inhibitor.4 When melanoma is diagnosed early, it is generally a curable disease,5,6 but most people with advanced melanoma have a poor prognosis.3 More than 232,000 people worldwide are currently diagnosed with melanoma each year.7 In recent years, there have been significant advances in treatment for metastatic melanoma, and people with the disease have more options. However, it continues to be a serious health issue with a high unmet need and a steadily increasing incidence over the past 30 years.8
About atezolizumab
Atezolizumab (also known as MPDL3280A) is an investigational monoclonal antibody designed to interfere with a protein called PD-L1. Atezolizumab is designed to target PD-L1 expressed on tumour cells and tumour-infiltrating immune cells, preventing it from binding to PD-1 and B7.1 on the surface of T cells. By inhibiting PD-L1, atezolizumab may enable the activation of T cells, restoring their ability to effectively detect and attack tumour cells.
About Zelboraf
Zelboraf was the first approved treatment for patients with unresectable or metastatic melanoma with BRAFV600 mutation as detected by a validated test, such as Roche's cobas 4800 BRAF Mutation Test. Zelboraf is not indicated for use in patients with wild-type BRAF melanoma.
About Cotellic plus Zelboraf
Cotellic and Zelboraf are used in combination to treat unresectable or metastatic melanoma with BRAFV600 mutation. Found in approximately half of melanomas, mutated BRAF causes abnormal signalling inside certain cancer cells leading to tumour growth. Zelboraf binds to mutant BRAF, to interrupt abnormal signalling that can cause tumours to grow.9,10Cotellic is designed to inhibit some forms of MEK. Both BRAF and MEK are proteins in a cell signalling pathway that help control cell growth and survival. When used in combination, Cotellic and Zelboraf are thought to reduce cancer cell growth longer than Zelboraf alone. A patient's healthcare provider will perform a test to make sure Cotellic and Zelboraf are right for the patient. It is not known if Cotellic and Zelboraf are safe and effective in children under 18 years of age.
Cotellic was approved in Switzerland in August 2015 and in the US in November 2015. In September, the Committee for Medicinal Products for Human Use (CHMP) at the European Medicines Agency (EMA) issued a positive opinion for Roche's marketing authorisation application for Cotellic in the European Union. A decision from the European Commission is expected before the end of 2015.
Roche in skin cancer
The Roche Group is the world's leading provider of cancer care products, including anti-cancer treatments, supportive care products and diagnostics. In the area of skin cancer, Roche scientists have been studying treatments for nearly 20 years. More than 28,000 patients having been treated worldwide, bringing about medical breakthroughs and new standards of care that include Zelboraf, Cotellic and Erivedge, treatments for two of the most difficult-to-treat skin cancers, metastatic melanoma and basal cell carcinoma. Roche is continuing to study skin cancer medicines as monotherapies and in combination with other investigational medicines, such as cancer immunotherapies, in several cancer types and diseases.
About Roche in cancer immunotherapy
For more than 30 years, Roche has been developing medicines with the goal to redefine treatment in oncology. Today, we're investing more than ever in our effort to bring innovative treatment options that help a person's own immune system fight cancer.
About personalised cancer immunotherapy
The aim of personalised cancer immunotherapy (PCI) is to provide individual patients with treatment options that are tailored to their specific needs. Our PCI research and development programme comprises more than 20 investigational candidates, eight of which are in clinical trials. All studies include the prospective evaluation of biomarkers to determine which people may be appropriate candidates for our medicines. In the case of atezolizumab (also known as MPDL3280A), PCI begins with the PD-L1 IHC assay based on the SP142 antibody developed by Roche. The goal of PD-L1 as a biomarker is to identify those who are most likely to experience clinical benefit with atezolizumab as a single agent and those who may be appropriate candidates for combination therapies; the purpose is not to exclude patients from atezolizumab therapy, but rather to enable the design of combinations that will provide the greatest chance for transformative responses. The ability to combine atezolizumab with multiple chemotherapies and targeted medicines may provide new treatment options to people across a broad range of tumours regardless of their level of PD-L1 expression.
About Roche
Headquartered in Basel, Switzerland, Roche is a leader in research-focused healthcare with combined strengths in pharmaceuticals and diagnostics. Roche is the world's largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and neuroscience. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management. Roche's personalised healthcare strategy aims at providing medicines and diagnostics that enable tangible improvements in the health, quality of life and survival of patients. Founded in 1896, Roche has been making important contributions to global health for more than a century. Twenty-nine medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and chemotherapy.
In 2014, the Roche Group employed 88,500 people worldwide, invested 8.9 billion Swiss francs in R&D and posted sales of 47.5 billion Swiss francs. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan. For more information, please visit www.roche.com.
All trademarks used or mentioned in this release are protected by law.
References
1.Hamid O, et al. Preliminary clinical safety, tolerability and activity of atezolizumab (anti-PDl1) combined with Zelboraf in BRAFv600 metastatic melanoma, PLENARY SESSION 5 - Melanoma and the Immune System. San Francisco Marriott Marquis, 9:35AM to 9:50AM
2.]. Friday 20th November, 09:35-09:50, San Francisco, United States.
3.Algazi AP, et al. Treatment of cutaneous melanoma: current approaches and future prospects. Cancer Manag Res. 2010;2:197–211.
4.Finn L, et al. Therapy for metastatic melanoma: the past, present, and future. BMC Med. 2012;10:23.
5.Ascierto PA, et al. The role of BRAF V600 mutation in melanoma. J Transl Med. 2012;10:85.
6.Leong SP. Future perspectives on malignant melanoma. Surg Clin North Am. 2003;83:453–6.
7.Creagan ET. Malignant melanoma: an emerging and preventable medical catastrophe. Mayo Clin Proc. 1997;72:570–4.
8.Ferlay J, et al. GLOBOCAN 2012 v1.0, Cancer incidence and mortality worldwide: IARC CancerBase No. 11. Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr/Pages/fact_sheets_population.aspx. Last accessed September 2015.
9.Bataille V. Risk factors for melanoma development. Expert Rev Dermatol. 2009;4:533–9.
10.Safaee Ardekani G, et al. The prognostic value of BRAF mutation in colorectal cancer and melanoma: a systematic review and meta-analysis. PLoS One. 2012;7(10):e47054.
11.Haferkamp S, et al. Zelboraf induces senescence features in melanoma cells. J Invest Dermatol. 2013;133:1601–9.