For years pharma skirted online message boards for fear of opening a Pandora's box of adverse event liabilities. But now academics are using the wealth of online medical talk to their advantage by searching for new areas of scientific investigation on message boards and Twitter.
The team at the University of Pennsylvania is sifting through anonymized online conversations to see what cancer patients say about herbal and nutritional supplements. If hundreds of patients are all talking about a particular health benefit or side effect, it could warrant closer attention. "It's useful to identify areas that would merit further study with all scientific rigor," Dr. John Holmes, an epidemiologist at the University of Pennsylvania, told The Wall Street Journal.
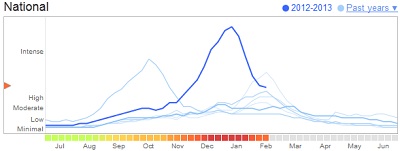 |
| Google tracks flu activity through searches.--Courtesy of Google |
The project comes amid growing interest in mining the internet for health trends and information. Last month researchers at Johns Hopkins University showed off their use of Twitter to track real-time flu trends. Google ($GOOG) has been tracking flu activity through searches for years and generally produces data that closely matches U.S. Centers for Disease Control and Prevention findings. In the recent flu season, however, Google majorly overestimated peak flu levels, Nature reports.
A few glitches are unlikely to dampen interest in tapping the power of online discussions, though. "We see patient conversations on the Internet as the largest post-marketing study ever," Michele Bennett, chief operating officer of Wool Labs, told the WSJ. Wool Labs runs a program to scour the internet looking for insight into patient beliefs and buying patterns for its pharma clients.
In 2010 Wool Labs showed the potential of its WebDig system by doing a retrospective analysis of online conversations about GlaxoSmithKline's ($GSK) diabetes drug Avandia. The FDA restricted use of Avandia in 2010, but WebDig showed that patients had begun to lose faith in the drug, GSK, their physicians and the regulator several years earlier.
- check out the WSJ feature (sub. req.)
- read the Nature blog on Google
- here's the Avandia study (PDF)